Novartis presents new Cosentyx data showing long-lasting efficacy in psoriatic arthritis over 3 years including patient-reported pain
· Cosentyx® data show sustained improvements in the signs and symptoms of psoriatic arthritis (PsA) in approximately 80% of patients over 3 years 1*
· New data show that with Cosentyx the high ACR response rates are consistently maintained in PsA, with no reduction from Year 1 to Year 31
· New head-to-head clinical trials planned in ankylosing spondylitis (AS) and PsA to compare Cosentyx versus Humira®2†‡
Basel, November 14, 2016 – Novartis announced today new data showing Cosentyx® (secukinumab) delivers sustained improvements in the signs and symptoms of psoriatic arthritis (PsA) over three years – including patient-reported pain1,3. These findings were presented at the 2016 Annual Meeting of the American College of Rheumatology (ACR) in Washington DC, United States, at which Novartis presented 28 abstracts in total.
Cosentyx is the first approved fully human interleukin-17A (IL-17A) inhibitor to demonstrate three-year efficacy in patients with PsA1,4, a life-long inflammatory disease that affects the skin and joints. Cosentyx is also the only IL-17A inhibitor indicated for ankylosing spondylitis (AS), PsA, and psoriasis4 – this is significant as up to eight in 10 patients diagnosed with PsA already have psoriasis5.
“Joint pain severely impacts the lives of many people living with psoriatic arthritis. The knowledge that Cosentyx delivers lasting pain relief, reduces swelling and helps keep joints moving is important as it means it could help improve both patient overall quality of life and mobility,” said Vasant Narasimhan, Global Head, Drug Development and Chief Medical Officer, Novartis. “Cosentyx provides much-needed pain relief and also delivers high and long-lasting skin clearance for psoriasis, which the majority of people with psoriatic arthritis also suffer from.”
In the first year of the three-year open-label extension study, which is a continuation of the two-year double-blind study previously reported, 77% of PsA patients achieved an ACR 20 response (American College of Rheumatology response criteria) with Cosentyx1*. Completion rates for the extension study were high with 95% patients completing the first year of the extension trial1. These new data show that response rates were consistent from Year 1 (69.4%) to Year 3 (76.8%) and this was independent of whether patients received an anti-TNF prior to receiving Cosentyx1. Importantly, a component of this measure includes patient-reported pain. Cosentyx has previously shown 79% of AS patients achieved an ASAS 20 response (Assessment of Spondyloarthritis International Society response criteria) at two years6§. Previous data also show up to 80% of AS and 84% of PsA patients treated with Cosentyx at two years had no radiographic progression in the spine or joints respectively, as shown by X-ray assessment7,8. Cosentyx continues to have a favorable safety profile, which was consistent with that shown in Phase III studies9-12.
In addition, analyses, using Matching-Adjusted Indirect Comparison (MAIC) presented at ACR show Cosentyx may improve the signs and symptoms of AS and PsA more than Humira® (adalimumab)†. In a new MAIC for AS, the ASAS 20 response rates at Week 52 were higher for Cosentyx (74%) than for Humira® (65%)13. Likewise, in the MAIC for PsA patients, ACR 20 response rates at Week 48 were higher for Cosentyx (72%) than for Humira® (56%)14.
Following these MAIC analyses, Novartis plans to initiate new head-to-head clinical trials to directly compare Cosentyx versus Humira® in patients with AS and PsA2‡. These will include 850 patients and will be the first ever adequately powered head-to-head studies with biologic medicines to differentiate the effectiveness of treatment in these conditions.
About Cosentyx and interleukin-17A (IL-17A)
Cosentyx is a fully human monoclonal antibody that selectively neutralizes IL-17A. Research suggests that IL-17A may play an important role in driving autoinflammatory conditions in enthesis and ultimately the body’s immune response in psoriasis, AS and PsA15,16.
Cosentyx is the first IL-17A inhibitor approved in more than 55 countries for the treatment of active AS and PsA, which includes the European Union countries, Switzerland, and the US. Cosentyx is also approved for the treatment of PsA and pustular psoriasis in Japan.
For patients with PsA in the US, Cosentyx may be administered with or without loading dosage by subcutaneous injection. With loading dosage, the recommended dose is Cosentyx 150 mg by subcutaneous injection with initial dosing at Weeks 0, 1, 2 and 3, followed by monthly maintenance dosing starting at Week 4. Without loading dosage, the recommended dose is 150 mg every 4 weeks. If a patient continues to have active PsA, a dosage of 300 mg should be considered. Cosentyx may be administered with or without methotrexate. For PsA patients with concomitant moderate-to-severe plaque psoriasis the dosing and administration recommendations for plaque psoriasis apply.
Cosentyx is approved in more than 65 countries for the treatment of moderate-to-severe plaque psoriasis which includes the European Union countries, Japan, Switzerland, Australia, the US and Canada. In Europe, Cosentyx is approved for the first-line systemic treatment of moderate-to-severe plaque psoriasis in adult patients4. In the US, Cosentyx is approved as a treatment for moderate-to-severe plaque psoriasis in adult patients who are candidates for systemic therapy or phototherapy (light therapy)17.
More than 10,000 patients have been treated with Cosentyx in clinical trial settings across multiple indications, and over 50,000 patients have been treated in the post-marketing setting2.
About the FUTURE 1 study
FUTURE 1 is a two-year, multi-center, randomized, placebo-controlled Phase III pivotal study to evaluate the efficacy of Cosentyx in patients with active PsA. FUTURE 1 enrolled 606 patients with active PsA and assessed Cosentyx with intravenous loading (10 mg/kg) and subcutaneous (75 mg and 150 mg) maintenance dosing. The primary endpoint assessed superiority of Cosentyx against placebo in the proportion of patients achieving the ACR 20 response at Week 2412. From Week 16, patients in the placebo arm of the study were re-randomized to receive Cosentyx 75 mg or 150 mg at either Week 16 or Week 24, based on clinical response. At Week 104, patients could enter the extension phase of the study1.
About MAIC and limitations of these studies
While these MAIC analyses have been performed using data from randomized controlled trials, there are limitations to these analyses due to differences in study designs and lower effective sample size for Cosentyx. In addition, only publicly available data has been used for Humira®. This impacts availability of comparative data and matching of patient populations in these studies. The PsA MAIC is based on 150 mg Cosentyx dosage. MAIC is a valid and accepted method for comparative effectiveness research18,19. Further details on the MAIC analyses are available in the presented scientific abstracts and posters18,19.
About ankylosing spondylitis and psoriatic arthritis
AS is part of a family of life-long inflammatory diseases that also includes PsA. It generally results in serious impairment of movement in the spine and physical function, which has an impact on quality of life. People in their teens and twenties, particularly males, are affected most often. Family members of those with AS are at higher risk20,21.
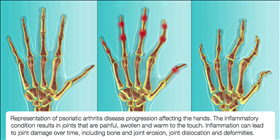
PsA is also closely associated with psoriasis. Approximately 30% of patients with psoriasis have PsA and as many as one in four people with psoriasis may have undiagnosed PsA22,23. Symptoms of PsA include joint pain and stiffness, skin and nail psoriasis, swollen toes and fingers, persistent painful swelling of the tendons, and irreversible joint damage5. Up to 40% of people can suffer from joint destruction and permanent physical deformity24.
About ASAS 20 and ACR 20
Improvements in the symptoms of AS are measured by the ASAS response criteria (ASAS 20), which is defined as an improvement of at least 20% and absolute improvement of at least 10 units on a 0-100mm scale in at least three of the following criteria: improvement in flexibility, night time pain, ability to perform specific tasks, morning stiffness, and no further deterioration in the condition. The percentage of patients reaching an ASAS 20 response is an accepted way of measuring the efficacy of treatments in AS25.
Improvements in the symptoms of PsA are measured by the ACR response criteria, which has three levels – ACR 20, 50, 70. In ACR 20 there is an improvement of 20% or greater of the following criteria: reduction in the number of tender joints; reduction in the number of swollen joints; and a reduction in three of five additional criteria, which are the patient’s assessment of pain and/or the patient’s assessment of how active his or her PsA is, the physician’s assessment of disease activity, levels of disability measured by the Stanford Health Assessment Questionnaire, and laboratory tests showing if inflammation is active. The ACR 50 and ACR 70 use the same criteria as ACR 20, but are looking for a higher percentage improvement (50% and 70%) instead of 20%26.
*In PsA 77% of patients achieved ACR 20 response; this includes both the anti-TNF naïve and anti-TNF inadequate response arms of the study.
†Humira is a registered trademark of AbbVie Inc.
‡In AS, a biosimilar of Humira® (adalimumab) will be used and in PsA the originator, Humira®, will be used to directly compare against Cosentyx.
§In AS, 79% of patients achieved ASAS 20 response; this includes both the anti-TNF naïve and anti-TNF inadequate response arms of the study.
Disclaimer
The foregoing release contains forward-looking statements that can be identified by words such as “planned,” “could,” “may,” “plans,” “will,” “suggests,” or similar terms, or by express or implied discussions regarding potential new indications or labeling for Cosentyx, or regarding potential future revenues from Cosentyx. You should not place undue reliance on these statements. Such forward-looking statements are based on the current beliefs and expectations of management regarding future events, and are subject to significant known and unknown risks and uncertainties. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those set forth in the forward-looking statements. There can be no guarantee that Cosentyx will be submitted or approved for any additional indications or labeling in any market, or at any particular time. Nor can there be any guarantee that Cosentyx will be commercially successful in the future. In particular, management’s expectations regarding Cosentyx could be affected by, among other things, the uncertainties inherent in research and development, including unexpected clinical trial results and additional analysis of existing clinical data; unexpected regulatory actions or delays or government regulation generally; the company’s ability to obtain or maintain proprietary intellectual property protection; general economic and industry conditions; global trends toward health care cost containment, including ongoing pricing pressures; unexpected safety, quality or manufacturing issues, and other risks and factors referred to in Novartis AG’s current Form 20-F on file with the US Securities and Exchange Commission. Novartis is providing the information in this press release as of this date and does not undertake any obligation to update any forward-looking statements contained in this press release as a result of new information, future events or otherwise.