Novartis’ Cosentyx superior to Stelara in delivering long-lasting skin clearance (PASI 90) for psoriasis patients at 52 weeks
· New data at AAD 2016 shows Cosentyx® is significantly more efficacious than Stelara in sustaining skin clearance (PASI 90 to PASI 100) at 52 weeks1
· Cosentyx delivered and sustained skin clearance (PASI 90 to PASI 100) in nearly
8 out of 10 moderate-to-severe psoriasis patients1
· Cosentyx granted first-line systemic indication for psoriasis in Europe and recently approved in psoriatic arthritis and ankylosing spondylitis in the US and Europe
Basel, March 5, 2016 – Novartis announced today new late-breaking data from the head-to-head CLEAR study, demonstrating that Cosentyx (secukinumab) remains superior to Stelara (ustekinumab) in achieving sustained skin clearance (PASI 90 response) at 52 weeks for adults living with moderate-to-severe psoriasis. These findings were presented for the first time at the American Academy of Dermatology (AAD) Annual Meeting in Washington, DC1.
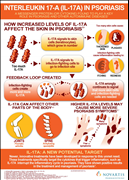
Cosentyx is the first fully human interleukin-17A inhibitor approved for adults to treat moderate-to-severe plaque psoriasis, and was recently approved for the treatment of psoriatic arthritis and ankylosing spondylitis in the EU and US.
“Cosentyx continues to demonstrate superior and sustainable efficacy against currently available biologics and is a proven first-line treatment option for adult patients with moderate-to-severe psoriasis,” said Vasant Narasimhan, Global Head, Drug Development and Chief Medical Officer, Novartis. “Cosentyx has the potential to give more people with psoriasis than ever before the benefit of long-lasting skin clearance.”
The ultimate aim of psoriasis treatment is clear skin, and the Psoriasis Area Severity Index (PASI) 90 response is considered an important measure of treatment success2,3. Meeting all primary and secondary endpoints at Weeks Four, 16 and 52, Cosentyx demonstrated it remains consistently superior to Stelara in achieving and sustaining PASI 90 response (76.2% vs. 60.6%; P<0.0001), and significantly better in achieving PASI 100 (clear skin) response (45.9% vs. 35.8%; P=0.0103) at 52 weeks. Cosentyx also showed significantly greater and sustained Dermatology Life Quality Index (DLQI) 0/1 responses versus Stelara (71.6% vs. 59.2%; P=0.0008)1.
The study also demonstrated Cosentyx had a superior rapid onset of action compared to Stelara, with half of Cosentyx patients achieving PASI 75 as early as Week Four (50.0% vs. 20.6%, P<0.0001)4. Cosentyx had a similar safety profile to that of Stelara in the study, which was consistent with that reported in the pivotal Cosentyx Phase III studies1.
Affecting around 125 million people globally, psoriasis is a chronic skin condition that causes itching, scaling and pain, and can have a significant impact on physical and psychological wellbeing5,6. Despite this, up to half of patients receive no treatment, and of those who do, many (52%) remain dissatisfied with their disease management7.
About psoriasis
Psoriasis is a common, non-contagious, autoimmune disease that affects up to 3% of the world’s population5. Plaque psoriasis is the most common form of the disease and appears as raised, red patches covered with a silvery white buildup of dead skin cells. Psoriasis is not simply a cosmetic problem, but a persistent, chronic (long-lasting), and sometimes distressing disease, which can affect even the smallest aspects of people’s lives on a daily basis. Up to 30% of patients with psoriasis have, or will, develop psoriatic arthritis, in which the joints are also affected, causing debilitating symptoms including pain, stiffness and irreversible joint damage8,9. Psoriasis is also associated with other serious health conditions, such as diabetes, heart disease and depression8.
About the CLEAR study
CLEAR (Comparison to assess Long-term Efficacy, sAfety and toleRability of secukinumab vs. ustekinumab) is a multi-center, double-blind, parallel-group study of Cosentyx (n=334) versus Stelara (n=335) to compare efficacy, safety and tolerability in adults with moderate-to-severe plaque psoriasis. Patients were randomized to receive either Cosentyx (300 mg) by subcutaneous injection at Baseline, Weeks One, Two and Three, then every four weeks from Week Four, or Stelara (dosing per package label). Cosentyx achieved the primary objective of superior PASI 90 response at Week 16. These data were published as an e-publication in the Journal of the American Academy of Dermatology, June 17, 20154. The 52-week PASI 90 response is a secondary objective in this study. PASI 100 and DLQI responses at 52 weeks are exploratory endpoints.
About Cosentyx and interleukin-17A (IL-17A)
Cosentyx is a fully human monoclonal antibody that selectively neutralizes circulating IL-17A. Research suggests that IL-17A may play an important role in driving the body’s immune response in psoriasis, psoriatic arthritis and ankylosing spondylitis10,11.
Cosentyx is approved in over 50 countries for the treatment of moderate-to-severe plaque psoriasis which includes the European Union countries, Japan, Switzerland, Australia, the US and Canada. In Europe, Cosentyx is the only biologic approved for the first-line systemic treatment of moderate-to-severe plaque psoriasis in adult patients. In the US, Cosentyx is approved as a treatment for moderate-to-severe plaque psoriasis in adult patients who are candidates for systemic therapy or phototherapy (light therapy).
In addition, Cosentyx is the first IL-17A inhibitor with positive Phase III results for the treatment of active psoriatic arthritis and active ankylosing spondylitis12-16 and is now approved in Europe, the US, Ecuador, Bangladesh and the Philippines for these conditions. Cosentyx is also approved for the treatment of psoriatic arthritis and pustular psoriasis in Japan.
Disclaimer
The foregoing release contains forward-looking statements that can be identified by words such as “continues,” “potential,” “aim,” “can,” “will,” “suggests,” “may,” or similar terms, or by express or implied discussions regarding potential new indications or labeling for Cosentyx, or regarding potential future revenues from Cosentyx. You should not place undue reliance on these statements. Such forward-looking statements are based on the current beliefs and expectations of management regarding future events, and are subject to significant known and unknown risks and uncertainties. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those set forth in the forward-looking statements. There can be no guarantee that Cosentyx will be submitted or approved for any additional indications or labeling in any market, or at any particular time. Nor can there be any guarantee that Cosentyx will be commercially successful in the future. In particular, management’s expectations regarding Cosentyx could be affected by, among other things, the uncertainties inherent in research and development, including unexpected clinical trial results and additional analysis of existing clinical data; unexpected regulatory actions or delays or government regulation generally; the company’s ability to obtain or maintain proprietary intellectual property protection; general economic and industry conditions; global trends toward health care cost containment, including ongoing pricing pressures; unexpected safety issues; unexpected manufacturing or quality issues, and other risks and factors referred to in Novartis AG’s current Form 20-F on file with the US Securities and Exchange Commission. Novartis is providing the information in this press release as of this date and does not undertake any obligation to update any forward-looking statements contained in this press release as a result of new information, future events or otherwise.