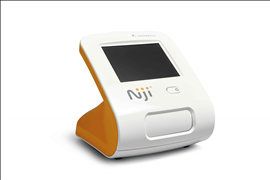
New Novartis point of care Niji™ System may provide earlier diagnosis of severe allergic asthma and faster treatment decisions
· NijiTM System, Total IgE Test delivers results rapidly, allowing for quick in-office diagnosis of IgE-mediated allergic disorders and faster treatment decisions
· Point of care in office diagnostics may accelerate treatment decisions that could lead to improved patient care and outcomes
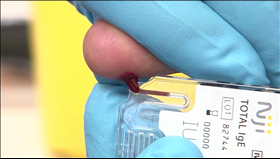
· Niji System, using only one to two droplets of finger stick blood, provides flexible and easy to use point of care platform with the potential to be used across various disease areas
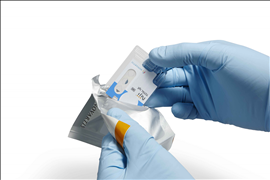
Basel, September 9, 2016 – Novartis announced today the introduction of a novel in office point of care diagnostic tool – the NijiTM System and Total IgE Test. This first test delivers quantitative total IgE (Immunoglobulin E) levels in about 12 minutes using only one to two droplets of finger stick blood, allowing for quick in-office diagnosis of IgE-mediated allergic disorders in conjunction with other clinical findings.
Identifying patients with uncontrolled asthma mediated by IgE remains an area of high unmet need. It is estimated that in Europe, more than 30 million people are affected by asthma, and approximately half of patients remain uncontrolled. Point of care testing in the healthcare provider’s office has the potential to reduce the time from consultation to diagnosis and to treatment decision, leading to improved patient outcomes. Current IgE tests are performed in central laboratories and it can take up to several weeks before results are known.
“Point of care testing is an important tool for healthcare professionals in order to make informed treatment decisions within a single appointment, thus helping to ensure patients are not lost to follow-up and ultimately improving patient management and outcomes,” said Vas Narasimhan, MD, Global Head of Drug Development and Chief Medical Officer for Novartis. “The Niji System provides a platform for fast and easy blood tests that could potentially be applied across a variety of disease areas.”
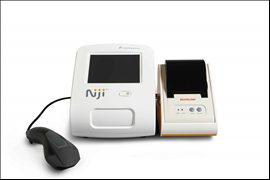
Novartis recently completed all requirements for the CE Mark of the point of care diagnostic platform NijiTM System as well as the Total IgE Test. The system is now cleared for sale within the European Union and in all countries recognizing the CE Mark and the company intends to launch the system in Europe in Q4 2016.
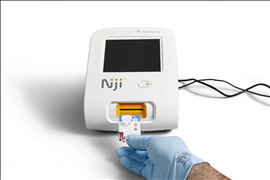
The NijiTM System does not require any sample preparation, lengthy setup, or any calibration procedures. Clinical and non-clinical performance evaluations conducted with the NijiTM Total IgE Test demonstrated performance comparable to currently-marketed reference laboratory tests. The test may be a useful tool for healthcare professionals as an aid in the diagnosis of IgE-mediated allergic disorders in conjunction with other clinical findings in their office setting.
About the NijiTM System
The NijiTM System is for use by medical professionals and consists of a small desk-top analyzer, optional accessories, and test-specific disposable cartridges. It is based on a proprietary pyro-electric technology that supports the design of sensitive, rapid immunoassay tests using unprocessed capillary blood specimens. This allows the development of tests which quantitate proteins, antibodies, drug molecules and metabolites in capillary blood and other body fluids within 10 to 15 minutes.
Novartis is currently developing tests on the NijiTM System in order to address unmet needs for diagnosis and management of patients, with a pipeline of several NijiTM tests in its portfolio. In addition to broad range of tests in development, Novartis is exploring partnerships to expand the menu of tests offered on the novel NijiTM System.
About Total IgE
Immunoglobulin E (IgE) and the total concentration in blood (Total IgE) is an important biomarker for the diagnosis and management of atopic asthma and other allergic conditions. Elevated IgE can be used as a first indicator for atopic asthma in the general population 1,2,3. In patients with asthma, higher IgE levels are associated with increased asthma severity and reduced lung function 4,5.
Disclaimer
The foregoing release contains forward-looking statements that can be identified by words such as “may,” “could,” “potential,” “introduction,” “potentially,” “cleared for sale,” “intends,” “launch,” “developing,” “pipeline,” “portfolio,” “exploring,” or similar terms, or by express or implied discussions regarding potential launches of the Niji System and Total IgE Test, other tests in development for the Niji System, or regarding potential future revenues from the Niji System, Total IgE Test and other tests in development for the Niji System. You should not place undue reliance on these statements. Such forward-looking statements are based on the current beliefs and expectations of management regarding future events, and are subject to significant known and unknown risks and uncertainties. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those set forth in the forward-looking statements. There can be no guarantee that the Niji System and Total IgE Test will be submitted or approved for sale in any additional markets, or at any particular time. Neither can there be any guarantee that the Niji System and Total IgE Test will be launched in any market, or at any particular time. Nor can there be any guarantee that any additional tests for the Niji System will be successfully developed, approved or launched, or at any particular time. Neither can there be any guarantee that the Niji System, Total IgE Test or any other test in development for the Niji system will be commercially successful in the future. In particular, management’s expectations regarding the Niji System, Total IgE Test and such other tests in development for the Niji System could be affected by, among other things, the uncertainties inherent in research and development, including unexpected clinical trial results and additional analysis of existing clinical data; unexpected regulatory actions or delays or government regulation generally; the company’s ability to obtain or maintain proprietary intellectual property protection; general economic and industry conditions; global trends toward health care cost containment, including ongoing pricing pressures; unexpected safety, quality or manufacturing issues, and other risks and factors referred to in Novartis AG’s current Form 20-F on file with the US Securities and Exchange Commission. Novartis is providing the information in this press release as of this date and does not undertake any obligation to update any forward-looking statements contained in this press release as a result of new information, future events or otherwise.