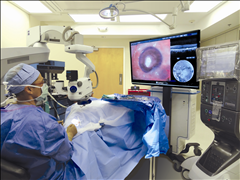
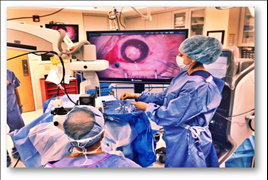
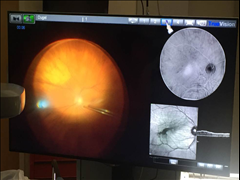
Alcon launches the NGENUITY® 3D Visualization System designed to further enhance retinal surgeon experience
· Heads-up design using high definition 3D screen engineered to improve surgeons posture and may reduce fatigue1
· Offers enhanced 3D visualization with high-definition resolution, image depth, clarity and color contrast while helping to minimize light exposure to the patient’s eye
· Vitreoretinal surgeries may range from thirty minutes to over three hours in length
Basel, Switzerland, September 8, 2016 – Alcon, the global leader in eye care and a division of Novartis, launches the NGENUITY® 3D Visualization System, a platform for Digitally Assisted Vitreoretinal Surgery (DAVS). The system is designed to enhance visualization of the back of the eye for improved surgeon experience. Alcon is launching the system in collaboration with TrueVision® 3D Surgical, a California-based company specializing in digital 3D visualization and guidance for microsurgery.
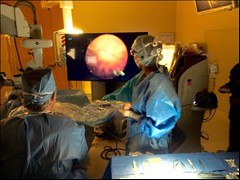
The NGENUITY® 3D Visualization System allows retinal surgeons to operate looking at a high definition 3D screen, instead of bending their necks to look through the eye-piece of a microscope. Traditional vitrectomy surgeries range from 30 minutes to over three hours in length to complete. This microscope-free design is engineered to improve surgeons’ posture and may reduce fatigue.1
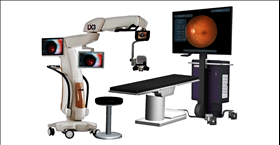
The NGENUITY 3D Visualization System is comprised of several elements, notably a High Dynamic Range (HDR) camera that provides excellent resolution, image depth, clarity and color contrast. With the three-dimensional view, the surgeon now has depth perception not previously available on standard television monitors, often used today in the operation theatre. Surgeons may also increase magnification while maintaining a wide field of view as well as use digital filters to customize his or her view during each procedure, highlighting ocular structures and tissue layers which is imperative to visualize the back of the eye. Engineered with a specific focus on minimizing light exposure to the patient’s eye, the NGENUITY 3D Visualization System facilitates operating using lower light levels.1
“This digital platform offers more than just enhanced visualization. It will impact our therapies and the way we manipulate tissue,” said Dr. Allen Ho, Professor of Ophthalmology, Thomas Jefferson University and Director of Retina Research Wills Eye Hospital, Philadelphia, PA, USA. “The easier it is for surgeons to perform these long, delicate surgeries, the better they can perform for our patients who count on us to provide them with the best possible care.”
The system is designed to facilitate collaboration and teaching in the operating room. Offering an immersive panoramic surgical view, the NGENUITY 3D Visualization System allows the operating team to see exactly what the surgeon is seeing in real-time.
“The NGENUITY 3D Visualization System takes vitreoretinal surgery to a more intuitive operating experience, offering greater depth and detail during surgery,” said Mike Ball, CEO of Alcon. “Our goal is to provide surgeons with better visualization while operating on the back of the eye, facilitate teaching and ultimately improve patient outcomes.”
The NGENUITY® 3D Visualization System is expected to be available in the United States and specific countries within Europe as of September 15, 2016, and is expected to be available in Japan in December of this year. Other countries are expected to launch the NGENUITY® 3D Visualization System over the course of 2017.
About Vitreoretinal Surgery2
Vitreoretinal surgery is a sub-specialty of ophthalmology focused in diseases and surgery of the back of the eye including the retina and the vitreous body of the eye. The retina is a light-sensitive area that includes the macula, which is made up of light-sensitive cells that provide sharp, detailed vision. The vitreous body of the eye is a clear gel that fills the space between the retina and the lens. The retina, the macula, and the vitreous body can all be subject to various diseases and conditions that can lead to blindness or vision loss and may require the attention of a vitreoretinal surgeon.
About NGENUITY
The NGENUITY® 3D Visualization System was developed in collaboration with TrueVision® 3D Surgical, a California-based leader in digital 3D visualization and guidance for microsurgery. It consists of a 3D stereoscopic, high-definition digital video camera and workstation to provide magnified stereoscopic images of objects during micro-surgery. It acts as an adjunct to the surgical microscope during surgery displaying real-time images or images from recordings. Please refer to the User Manual for a complete list of appropriate uses, warnings and precautions.
Disclaimer
The foregoing release contains forward-looking statements that can be identified by words such as “launches,” “designed to,” “engineered to,” “may,” “offers,” “launching,” “will,” “offering,” “goal,” “ultimately,” “expected,” “can,” or similar terms, or by express or implied discussions regarding potential marketing approvals and launches of the NGENUITY 3D Visualization System, or regarding potential future revenues from the NGENUITY 3D Visualization System. You should not place undue reliance on these statements. Such forward-looking statements are based on the current beliefs and expectations of management regarding future events, and are subject to significant known and unknown risks and uncertainties. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those set forth in the forward-looking statements. There can be no guarantee that the NGENUITY 3D Visualization System will be submitted or approved for sale in any market, or at any particular time. Neither can there be any guarantee that the NGENUITY 3D Visualization System will be launched in any market, or at any particular time. Nor can there be any guarantee that the NGENUITY 3D Visualization System will be commercially successful in the future. In particular, management’s expectations regarding the NGENUITY 3D Visualization System could be affected by, among other things, the uncertainties inherent in research and development, including unexpected clinical trial results and additional analysis of existing clinical data; unexpected regulatory actions or delays or government regulation generally; the company’s ability to obtain or maintain proprietary intellectual property protection; general economic and industry conditions; global trends toward health care cost containment, including ongoing pricing pressures; unexpected safety, quality or manufacturing issues, and other risks and factors referred to in Novartis AG’s current Form 20-F on file with the US Securities and Exchange Commission. Novartis is providing the information in this press release as of this date and does not undertake any obligation to update any forward-looking statements contained in this press release as a result of new information, future events or otherwise.